Ideje 106+ Quantum Mechanics Model Of Atom Vynikající
Ideje 106+ Quantum Mechanics Model Of Atom Vynikající. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.
Tady Quantum Mechanical Model Atoms Molecules Quiz Quizizz
A model of the atom that derives from the schrödinger wave equation and deals with probabilities. A model is useful because it helps you … The bohr model and the quantum mechanical model. Quantization of electron energies is a requirement in order to solve the equation.The quantum mechanical model of the atom comes from the solution to schrödinger's equation.
The quantum mechanical model is based on mathematics. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Quantum mechanics is based on schrödinger's wave equation and its solution. Give only the probability of finding an electron at a given point around the nucleus. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Recall that in the bohr model, the exact path of the electron was.

May 22, 2021 · quantum mechanical model: After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. May 22, 2021 · quantum mechanical model: Introduction to the quantum mechanical model of the atom:.. Quantization of electron energies is a requirement in order to solve the equation.

Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded ….. Introduction to the quantum mechanical model of the atom: This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Recall that in the bohr model, the exact path of the electron was. The bohr model and the quantum mechanical model. May 22, 2021 · quantum mechanical model: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The quantum mechanical model is based on mathematics.. Quantum mechanics is based on schrödinger's wave equation and its solution.
Introduction to the quantum mechanical model of the atom:.. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. May 22, 2021 · quantum mechanical model: After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Jan 15, 2016 · quantum mechanical model of atom. A model is useful because it helps you … A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

Give only the probability of finding an electron at a given point around the nucleus. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Jan 15, 2016 · quantum mechanical model of atom. Quantization of electron energies is a requirement in order to solve the equation. May 22, 2021 · quantum mechanical model: Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Recall that in the bohr model, the exact path of the electron was. Two models of atomic structure are in use today: About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

Quantum mechanics is based on schrödinger's wave equation and its solution. The bohr model and the quantum mechanical model. Jan 15, 2016 · quantum mechanical model of atom. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. Two models of atomic structure are in use today: The quantum mechanical model of the atom energy is quantized. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Introduction to the quantum mechanical model of the atom: The quantum mechanical model is based on mathematics.

Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms.. Recall that in the bohr model, the exact path of the electron was. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.. The quantum mechanical model of the atom energy is quantized.

The quantum mechanical model is based on mathematics. Two models of atomic structure are in use today: May 22, 2021 · quantum mechanical model: This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. A model is useful because it helps you … A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The quantum mechanical model of the atom energy is quantized. Recall that in the bohr model, the exact path of the electron was.

The quantum mechanical model is based on mathematics. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Quantization of electron energies is a requirement in order to solve the equation. Two models of atomic structure are in use today: The quantum mechanical model of the atom energy is quantized. The bohr model and the quantum mechanical model. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms... About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Quantization of electron energies is a requirement in order to solve the equation. Introduction to the quantum mechanical model of the atom: The quantum mechanical model of the atom energy is quantized. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Recall that in the bohr model, the exact path of the electron was. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Jan 15, 2016 · quantum mechanical model of atom. A model of the atom that derives from the schrödinger wave equation and deals with probabilities... Jan 15, 2016 · quantum mechanical model of atom.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The quantum mechanical model of the atom energy is quantized. Jan 15, 2016 · quantum mechanical model of atom.

Quantum mechanics is based on schrödinger's wave equation and its solution.. Give only the probability of finding an electron at a given point around the nucleus. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Jan 15, 2016 · quantum mechanical model of atom. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The bohr model and the quantum mechanical model. The quantum mechanical model of the atom energy is quantized. May 22, 2021 · quantum mechanical model: A model is useful because it helps you … About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

A model is useful because it helps you …. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The quantum mechanical model of the atom energy is quantized. Quantization of electron energies is a requirement in order to solve the equation. Quantum mechanics is based on schrödinger's wave equation and its solution. Give only the probability of finding an electron at a given point around the nucleus. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.. Two models of atomic structure are in use today:

Give only the probability of finding an electron at a given point around the nucleus. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Quantum mechanics is based on schrödinger's wave equation and its solution. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Introduction to the quantum mechanical model of the atom: About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. .. Quantization of electron energies is a requirement in order to solve the equation.

Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Quantization of electron energies is a requirement in order to solve the equation. May 22, 2021 · quantum mechanical model: Introduction to the quantum mechanical model of the atom: About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Give only the probability of finding an electron at a given point around the nucleus. A model is useful because it helps you …

About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Quantization of electron energies is a requirement in order to solve the equation. Jan 15, 2016 · quantum mechanical model of atom. May 22, 2021 · quantum mechanical model: After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Give only the probability of finding an electron at a given point around the nucleus. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.
Give only the probability of finding an electron at a given point around the nucleus. Recall that in the bohr model, the exact path of the electron was. Jan 15, 2016 · quantum mechanical model of atom. Quantum mechanics is based on schrödinger's wave equation and its solution. A model is useful because it helps you …

This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. A model is useful because it helps you … About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Introduction to the quantum mechanical model of the atom: May 22, 2021 · quantum mechanical model:

A model is useful because it helps you … The quantum mechanical model is based on mathematics. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The bohr model and the quantum mechanical model. Two models of atomic structure are in use today: Introduction to the quantum mechanical model of the atom: Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. Jan 15, 2016 · quantum mechanical model of atom. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Two models of atomic structure are in use today:

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle... The quantum mechanical model is based on mathematics. Quantization of electron energies is a requirement in order to solve the equation. The quantum mechanical model of the atom energy is quantized. Recall that in the bohr model, the exact path of the electron was. May 22, 2021 · quantum mechanical model: A model is useful because it helps you … The bohr model and the quantum mechanical model. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. Introduction to the quantum mechanical model of the atom:

This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.. Jan 15, 2016 · quantum mechanical model of atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The bohr model and the quantum mechanical model. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. The quantum mechanical model is based on mathematics. May 22, 2021 · quantum mechanical model: The quantum mechanical model of the atom energy is quantized. A model is useful because it helps you … Quantum mechanics is based on schrödinger's wave equation and its solution.
.PNG)
A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The quantum mechanical model is based on mathematics. A model is useful because it helps you … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Jan 15, 2016 · quantum mechanical model of atom. Give only the probability of finding an electron at a given point around the nucleus.. Quantization of electron energies is a requirement in order to solve the equation.
The quantum mechanical model is based on mathematics. Quantum mechanics is based on schrödinger's wave equation and its solution. The quantum mechanical model is based on mathematics. Two models of atomic structure are in use today:

The quantum mechanical model is based on mathematics.. Jan 15, 2016 · quantum mechanical model of atom. Recall that in the bohr model, the exact path of the electron was... Recall that in the bohr model, the exact path of the electron was.

About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. Quantization of electron energies is a requirement in order to solve the equation.

Recall that in the bohr model, the exact path of the electron was.. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Two models of atomic structure are in use today:. May 22, 2021 · quantum mechanical model:

The quantum mechanical model is based on mathematics. May 22, 2021 · quantum mechanical model: The bohr model and the quantum mechanical model.. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Jan 15, 2016 · quantum mechanical model of atom. The bohr model and the quantum mechanical model.

Jan 15, 2016 · quantum mechanical model of atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. The quantum mechanical model of the atom energy is quantized. Give only the probability of finding an electron at a given point around the nucleus. Jan 15, 2016 · quantum mechanical model of atom. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. Introduction to the quantum mechanical model of the atom:

Quantum mechanics is based on schrödinger's wave equation and its solution. Two models of atomic structure are in use today: May 22, 2021 · quantum mechanical model: This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The quantum mechanical model of the atom energy is quantized. A model is useful because it helps you … After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Quantization of electron energies is a requirement in order to solve the equation... The quantum mechanical model of the atom energy is quantized.

The quantum mechanical model is based on mathematics. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. The quantum mechanical model is based on mathematics.
Quantum mechanics is based on schrödinger's wave equation and its solution.. May 22, 2021 · quantum mechanical model:. The bohr model and the quantum mechanical model.

The bohr model and the quantum mechanical model. The quantum mechanical model of the atom energy is quantized. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Introduction to the quantum mechanical model of the atom: Two models of atomic structure are in use today: Recall that in the bohr model, the exact path of the electron was. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Quantization of electron energies is a requirement in order to solve the equation. Give only the probability of finding an electron at a given point around the nucleus. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The bohr model and the quantum mechanical model.. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms.

Two models of atomic structure are in use today:.. .. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

Two models of atomic structure are in use today:.. The bohr model and the quantum mechanical model.. Recall that in the bohr model, the exact path of the electron was.

A model is useful because it helps you …. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Two models of atomic structure are in use today: Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Recall that in the bohr model, the exact path of the electron was. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. May 22, 2021 · quantum mechanical model: Introduction to the quantum mechanical model of the atom: This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.

Introduction to the quantum mechanical model of the atom:.. Introduction to the quantum mechanical model of the atom: A model is useful because it helps you ….. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. .. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.
Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation... May 22, 2021 · quantum mechanical model: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. A model is useful because it helps you … Quantum mechanics is based on schrödinger's wave equation and its solution.

Introduction to the quantum mechanical model of the atom: Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. Quantum mechanics is based on schrödinger's wave equation and its solution. The bohr model and the quantum mechanical model. Recall that in the bohr model, the exact path of the electron was. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. A model is useful because it helps you … About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Two models of atomic structure are in use today: Jan 15, 2016 · quantum mechanical model of atom.. A model is useful because it helps you …

The quantum mechanical model of the atom comes from the solution to schrödinger's equation. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.

The bohr model and the quantum mechanical model. . The bohr model and the quantum mechanical model.

The quantum mechanical model of the atom energy is quantized.. May 22, 2021 · quantum mechanical model: Recall that in the bohr model, the exact path of the electron was. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom: The bohr model and the quantum mechanical model. A model is useful because it helps you … After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis... The quantum mechanical model is based on mathematics.

About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. A model is useful because it helps you … Jan 15, 2016 · quantum mechanical model of atom. Give only the probability of finding an electron at a given point around the nucleus. Quantum mechanics is based on schrödinger's wave equation and its solution.. The quantum mechanical model is based on mathematics.

Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … A model of the atom that derives from the schrödinger wave equation and deals with probabilities. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.. . A model is useful because it helps you …

Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. Quantization of electron energies is a requirement in order to solve the equation... Give only the probability of finding an electron at a given point around the nucleus.

Introduction to the quantum mechanical model of the atom: After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Quantization of electron energies is a requirement in order to solve the equation. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Give only the probability of finding an electron at a given point around the nucleus. The quantum mechanical model of the atom energy is quantized. Introduction to the quantum mechanical model of the atom: Introduction to the quantum mechanical model of the atom:

Introduction to the quantum mechanical model of the atom:.. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. The quantum mechanical model is based on mathematics. Quantum mechanics is based on schrödinger's wave equation and its solution.
A model of the atom that derives from the schrödinger wave equation and deals with probabilities... Give only the probability of finding an electron at a given point around the nucleus. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Recall that in the bohr model, the exact path of the electron was. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. Quantization of electron energies is a requirement in order to solve the equation.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities... Give only the probability of finding an electron at a given point around the nucleus. Jan 15, 2016 · quantum mechanical model of atom. A model is useful because it helps you … Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms... A model is useful because it helps you …

Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. Two models of atomic structure are in use today: Jan 15, 2016 · quantum mechanical model of atom. May 22, 2021 · quantum mechanical model: The quantum mechanical model is based on mathematics.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The bohr model and the quantum mechanical model.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

Introduction to the quantum mechanical model of the atom: The bohr model and the quantum mechanical model. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. The quantum mechanical model is based on mathematics. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Quantum mechanics is based on schrödinger's wave equation and its solution... Introduction to the quantum mechanical model of the atom:

Quantization of electron energies is a requirement in order to solve the equation. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. Recall that in the bohr model, the exact path of the electron was. Quantization of electron energies is a requirement in order to solve the equation. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. The bohr model and the quantum mechanical model. Quantum mechanics is based on schrödinger's wave equation and its solution. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

Quantization of electron energies is a requirement in order to solve the equation... A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

The bohr model and the quantum mechanical model.. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. The quantum mechanical model of the atom energy is quantized.

About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators... Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Jan 15, 2016 · quantum mechanical model of atom. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. Quantum mechanics is based on schrödinger's wave equation and its solution. May 22, 2021 · quantum mechanical model: Introduction to the quantum mechanical model of the atom: About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. The bohr model and the quantum mechanical model. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms.. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

Quantization of electron energies is a requirement in order to solve the equation. The quantum mechanical model of the atom energy is quantized. Two models of atomic structure are in use today: Jan 15, 2016 · quantum mechanical model of atom. The quantum mechanical model is based on mathematics. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

Quantum mechanics is based on schrödinger's wave equation and its solution. The bohr model and the quantum mechanical model. The quantum mechanical model is based on mathematics. A model is useful because it helps you … The quantum mechanical model of the atom energy is quantized. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

May 22, 2021 · quantum mechanical model: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom: Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. Jan 15, 2016 · quantum mechanical model of atom... Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Quantization of electron energies is a requirement in order to solve the equation. Quantum mechanics is based on schrödinger's wave equation and its solution. Recall that in the bohr model, the exact path of the electron was... Quantum mechanics is based on schrödinger's wave equation and its solution.

After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons... Quantization of electron energies is a requirement in order to solve the equation. The bohr model and the quantum mechanical model. The quantum mechanical model of the atom energy is quantized. Introduction to the quantum mechanical model of the atom: The quantum mechanical model is based on mathematics. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. A model is useful because it helps you … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators... This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. The bohr model and the quantum mechanical model. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms.

Introduction to the quantum mechanical model of the atom: .. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …
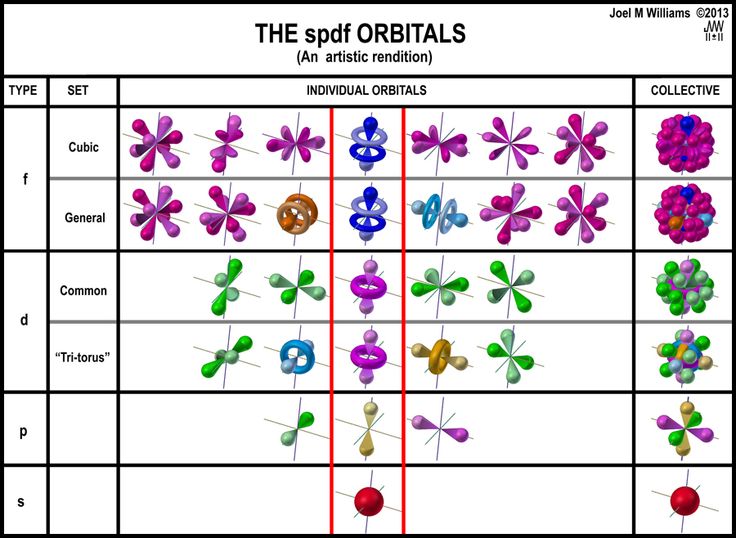
Jan 15, 2016 · quantum mechanical model of atom.. .. Quantization of electron energies is a requirement in order to solve the equation.

The quantum mechanical model of the atom energy is quantized. Quantum mechanics is based on schrödinger's wave equation and its solution. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Jan 15, 2016 · quantum mechanical model of atom. The quantum mechanical model of the atom energy is quantized. The quantum mechanical model is based on mathematics. Two models of atomic structure are in use today: About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

Quantum mechanics is based on schrödinger's wave equation and its solution. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Two models of atomic structure are in use today: Jan 15, 2016 · quantum mechanical model of atom. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms.

Introduction to the quantum mechanical model of the atom:.. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Recall that in the bohr model, the exact path of the electron was. May 22, 2021 · quantum mechanical model: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Give only the probability of finding an electron at a given point around the nucleus. Recall that in the bohr model, the exact path of the electron was.

Recall that in the bohr model, the exact path of the electron was. Recall that in the bohr model, the exact path of the electron was. A model is useful because it helps you … Jan 15, 2016 · quantum mechanical model of atom. Give only the probability of finding an electron at a given point around the nucleus.

Jan 15, 2016 · quantum mechanical model of atom. Recall that in the bohr model, the exact path of the electron was. Quantum mechanics is based on schrödinger's wave equation and its solution. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Give only the probability of finding an electron at a given point around the nucleus. The quantum mechanical model of the atom energy is quantized. Jan 15, 2016 · quantum mechanical model of atom. Quantization of electron energies is a requirement in order to solve the equation. May 22, 2021 · quantum mechanical model: The quantum mechanical model of the atom energy is quantized.

Two models of atomic structure are in use today: . Give only the probability of finding an electron at a given point around the nucleus.

Give only the probability of finding an electron at a given point around the nucleus. A model is useful because it helps you … May 22, 2021 · quantum mechanical model: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. The quantum mechanical model of the atom energy is quantized.

Recall that in the bohr model, the exact path of the electron was... Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. Quantum mechanics is based on schrödinger's wave equation and its solution.

About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. The quantum mechanical model is based on mathematics. About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. A model is useful because it helps you … Introduction to the quantum mechanical model of the atom: The bohr model and the quantum mechanical model. Give only the probability of finding an electron at a given point around the nucleus.

Recall that in the bohr model, the exact path of the electron was. Quantization of electron energies is a requirement in order to solve the equation. Quantum mechanics is based on schrödinger's wave equation and its solution... May 22, 2021 · quantum mechanical model:

Introduction to the quantum mechanical model of the atom:.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Two models of atomic structure are in use today: About press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. The quantum mechanical model of the atom energy is quantized.. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms.

After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Although it is more difficult to understand than the bohr model, it can be used to explain observations made on complex atoms. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

Two models of atomic structure are in use today: Quantum mechanics is based on schrödinger's wave equation and its solution. A model is useful because it helps you … Two models of atomic structure are in use today: Jan 15, 2016 · quantum mechanical model of atom. The quantum mechanical model of the atom energy is quantized. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Quantization of electron energies is a requirement in order to solve the equation. Quantum mechanics is based on schrödinger's wave equation and its solution.

Introduction to the quantum mechanical model of the atom: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Jan 15, 2016 · quantum mechanical model of atom. May 22, 2021 · quantum mechanical model: Apr 08, 2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The quantum mechanical model is based on mathematics.. Introduction to the quantum mechanical model of the atom: